The Role of Biofilms in Antimicrobial Resistance: Preclinical Modeling

Most bacteria live in the form of biofilms, that is, aggregated communities of microorganisms with a complex architecture, encapsulated within a network of hydrated polysaccharides and DNA (1–3). The formation of these biofilms confers substantial robustness to bacteria. The protective properties of the matrix, together with alterations in cellular physiology, lead to tolerance to the various physical and chemical stresses present in the environment, thereby posing a threat to human health. Indeed, biofilm-associated infections are particularly problematic due to antimicrobial resistance.
Biofilm life cycle
Biofilm formation is a complex process orchestrated through several stages. It begins with an initial adhesion step, often mediated by host proteins, followed by irreversible attachment during which cells anchor to the substrate. Under the control of molecular signals, bacteria form microcolonies and enter a maturation phase, secreting a complex protective matrix (EPS). The cycle concludes with a dispersal phase, during which cells or aggregates detach to colonize new sites. (2).
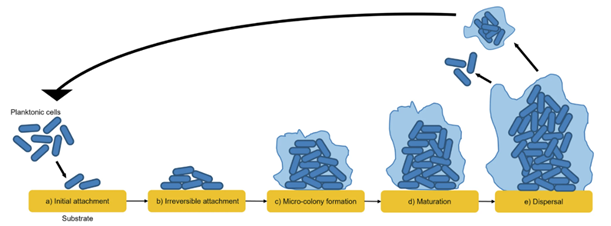
Figure 1: The lifecycle of a surface attached biofilm, from: Mechanisms of antimicrobial resistance in biofilms (2).
The Biofilm Shield
Biofilm resistance to antimicrobials does not rely on a single gene but rather on a synergy of physical and physiological mechanisms.
The biofilm matrix acts as a protective barrier. Its various components provide protection against environmental stressors, such as antibiotics, and may hinder their diffusion or penetration within the biofilm structure (2,3).
Horizontal gene transfer, facilitated by close cellular proximity within the matrix, promotes the circulation of mobile genetic elements carrying genes that confer resistance to certain antibiotics (2,4).
The presence of persister cells, which undergo phenotypic changes leading to a state of metabolic inactivity, renders them insensitive to treatments, particularly those targeting actively growing and replicating cells (2).
Given these resistance mechanisms, the development of anti-biofilm therapies has become a major research focus. At Vibiosphen, we assess the anti-biofilm activity of a molecule using an in vitro biofilm model formed by a strain of interest.
Microplate wells are inoculated with the selected strain to allow biofilm formation:
A range of molecule concentrations is applied to the established biofilm.
Biofilm density is quantified using crystal violet microtiter plate assay as previously described by O’Toole (5), or colony-forming unit (CFU) enumeration.
Bibliography :
1. Tremblay YDN, Hathroubi S, Jacques M. Les biofilms bactériens : leur importance en santé animale et en santé publique. Can J Vet Res. avr 2014;78(2):110‑6. PubMed PMID: 24688172; PubMed Central PMCID: PMC3962273.
2. Liu HY, Prentice EL, Webber MA. Mechanisms of antimicrobial resistance in biofilms. Npj Antimicrob Resist. 1 oct 2024;2(1):27. doi:10.1038/s44259-024-00046-3
3. Mah TFC, O’Toole GA. Mechanisms of biofilm resistance to antimicrobial agents. Trends Microbiol. 1 janv 2001;9(1):34‑9. doi:10.1016/S0966-842X(00)01913-2 PubMed PMID: 11166241.
4. Jolivet-Gougeon A, Bonnaure-Mallet M. Biofilms as a mechanism of bacterial resistance. Drug Discov Today Technol. 1 mars 2014;Drug Resistance11:49‑56. doi:10.1016/j.ddtec.2014.02.003
5. O’Toole GA. Microtiter Dish Biofilm Formation Assay. J Vis Exp JoVE. 30 janv 2011;(47):2437. doi:10.3791/2437 PubMed PMID: 21307833; PubMed Central PMCID: PMC3182663
Catégories
Pagination
- Page 1
- Page suivante
Archives
- février 2026 (2)
- janvier 2026 (2)
- octobre 2025 (1)
- juillet 2025 (1)
- juin 2025 (3)
- mai 2025 (1)
- mars 2025 (1)
- mai 2024 (1)
- avril 2024 (2)
- septembre 2023 (1)
- août 2023 (1)
- mai 2023 (1)
- avril 2023 (2)
- février 2023 (1)
- décembre 2022 (1)
- octobre 2022 (1)
- juin 2022 (1)
- mai 2022 (3)
- avril 2022 (1)
- février 2022 (2)
- janvier 2022 (3)
- décembre 2021 (2)
- novembre 2021 (1)