Determination of cytotoxicity
In vitro testing Cellular assays
Background of cytotoxicity
Determining the cytotoxic activity of an ingredient or molecule in preclinical studies is done to evaluate its safety profile.
By testing a new compound on various cell lines (e.g., liver, kidney, or heart cells), highly toxic molecules can quickly be identified.
Using different cell types, one can predict which organs might be susceptible to damage. For example, a compound that is highly cytotoxic to a liver cell line might indicate a risk of hepatotoxicity in a living organism.
These assays help establish the concentration at which a compound becomes toxic, often expressed as the CC₅₀ (half maximal cytotoxic concentration).
This data is essential for setting a safe starting dose range for animal studies and, eventually, human clinical trials.
Objectives of the model
• To characterize the cytotoxic activity of compounds when applied on eukaryotic cells.
Our approach at Vibiosphen
• Our library of cell lines
Immortalized or primary cells lines can be used. Additional cell lines can be purchased upon request.
• Method
6 concentrations of 5 compounds were applied to cell monolayers in duplicate.
The impact of the vehicle was also investigated.
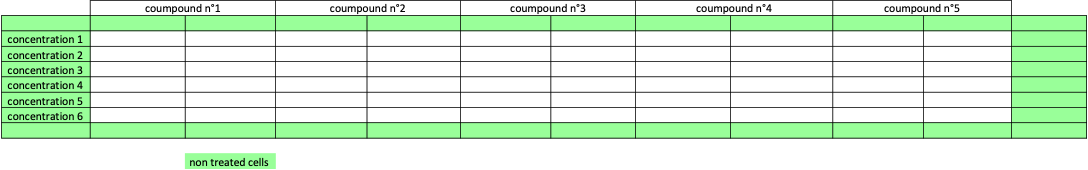
Plate design-example
The plate was incubated at 37°C.
Viable cells were quantified with Cell Titer Glo 2.0 assay.
Outcomes of Cytotoxicity assay

Table-Cell percentage survival. Normalized to 100% cell viability in non treated cells
Compound n°2 and n°5 were cytotoxic at 600µg/ml.
Compound n°4 was cytotoxic from 600 to 200µg/ml.
Compound n°1 and n°3 were not cytotoxic for all the tested concentrations.
