Determination of Minimum inhibitory concentration (MIC)
In vitro testing Microbiology powered
Background of Minimum inhibitory concentration (MIC)
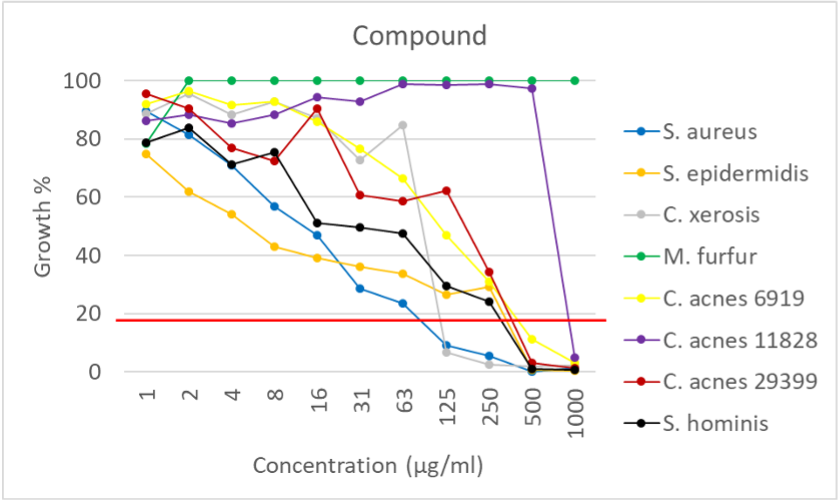
The minimum inhibitory concentration (MIC) is the lowest concentration of an antimicrobial that prevents visible growth of a microorganism.
Determining the MIC for any new antimicrobial is a fundamental step in its development:
- MIC value provides a quantitative measure of an antimicrobial's effectiveness
- MIC is a critical parameter for determining the appropriate dosage for a drug. To be effective, the concentration of the drug at the site of infection must exceed the MIC for a sufficient period of time.
EUCAST (European Committee on Antimicrobial Susceptibility Testing) provides guidance for performing and interpreting antimicrobial (bacteria and fungi) susceptibility testing. This ensures that test results are reliable and comparable across different laboratories.
Objectives of the model
- To determine the MIC of antibiotics and antifungals in development
Our approach at Vibiosphen
Our library of bacterial and fungal species

Reference strains and clinical isolates are available. Additional strains can be purchased upon request.
Method
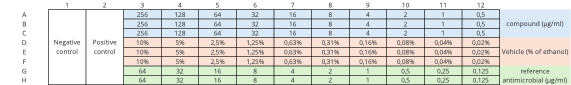
The standardized broth microdilution method provided by EUCAST is applied.
The three main controls used are:
- the growth control to confirm that the test conditions (medium's quality, temperature, and inoculum viability) are suitable for the organism to grow
- the sterility control should show no visible growth after incubation
- the reference antimicrobial control to ensure that the accuracy and reproducibility of the MIC test itself.
Outcomes of Minimum inhibitory concentration (MIC)